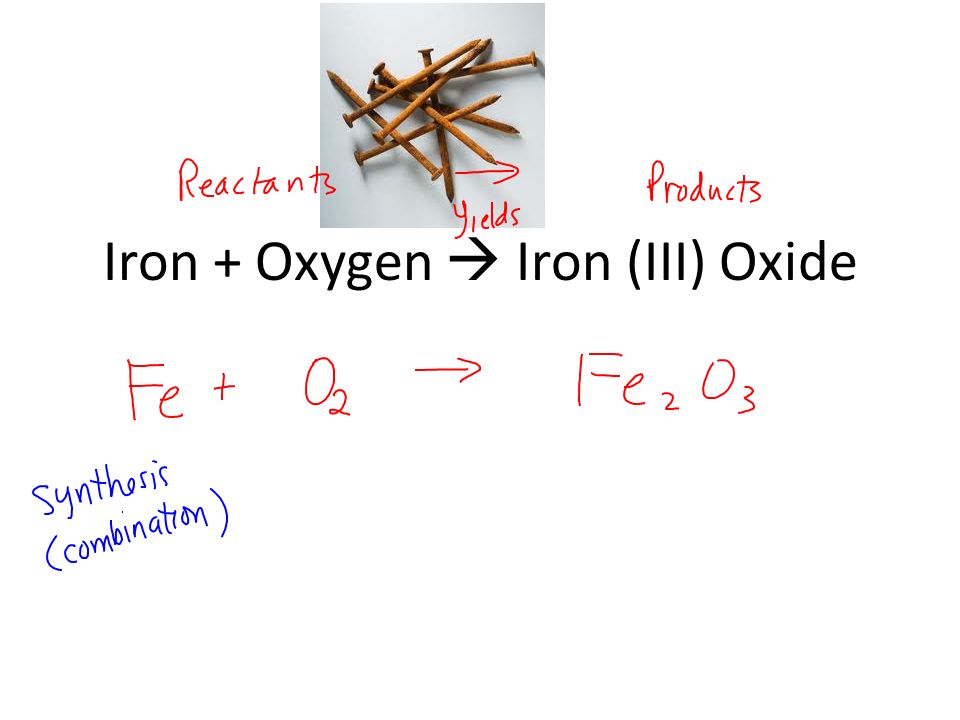
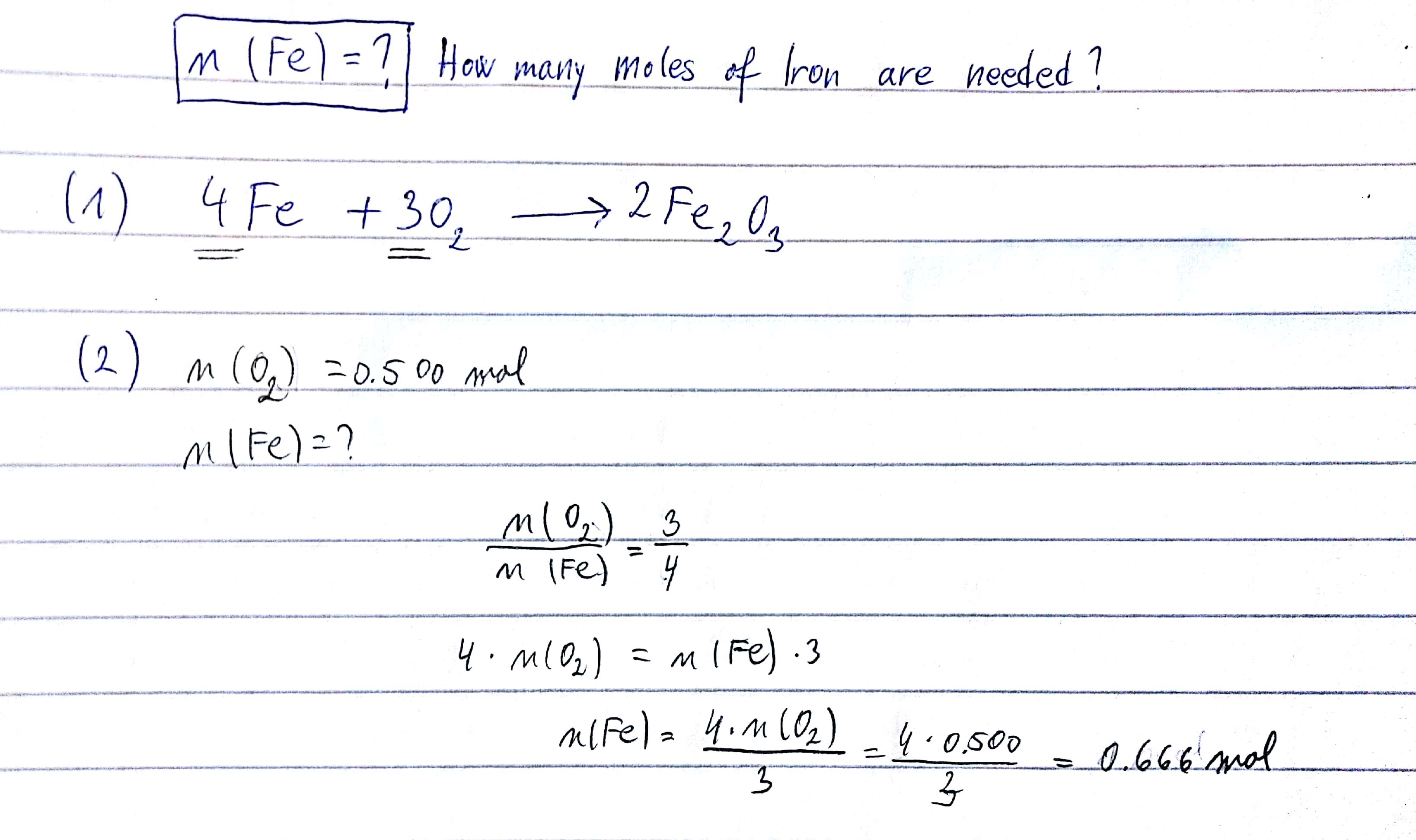Vinegar And Clr Are My New Best Friends - Iron Rusting Chemical Equation PNG Image | Transparent PNG Free Download on SeekPNG

Types of Chemical Reactions 8-2 Beaker Breaker Balance the following equation: ______H 3 PO 4 _______H 4 P 2 O 7 + ______H 2 O. - ppt download

Simplified chemical reactions between iron and oxygen within the haem... | Download Scientific Diagram

Causes chemistry of rusting rust prevention introduction to oxidation reduction REDOX reactions gcse igcse KS4 science chemistry revision notes revising














:max_bytes(150000):strip_icc()/BalanceEquations1-56a132765f9b58b7d0bcf535.png)